One place for your practice and patients, every step of the way
Personalized education and customized treatment support through a single point of engagement
Prescribe and connect your patients to myAgios® Patient Support Services
Prescribe onlineHaving trouble? Download the form instead.
You can also get started by calling us at 1-877-77-AGIOS (1-877-772-4467), Mon-Fri, 8 AM to 8 PM ET.
Starting AQVESME
All-in-one enrollment into myAgios and support for your patients
myAgios offers centralized support for your office staff when seeking care for your patients living with thalassemia. AQVESME has a Risk Evaluation and Mitigation Strategy (REMS) to help ensure safe use, regulatory compliance, and coordinated care for patients. Below, you'll find information and resources on how to get your patients started with AQVESME.
Personalized Patient Education
Every patient's treatment experience is unique. Our Agios Clinical Educators (ACEs) will share information that is tailored to each patient's individual needs, providing education and support pre- and post-prescription.
Support Through
Enrollment
A one-time AQVESME REMS certification is required for prescribers (AQVESMEREMS.com).
Before treatment initiation:
- Counsel the patient
- Obtain liver tests to establish a baseline
- Complete 2 forms
Access and Assistance
Patient Support Managers (PSMs) partner with your patients and your practice to facilitate access to AQVESME. They will help coordinate insurance and delivery through the exclusive REMS-certified Specialty Pharmacy.
Customized Treatment
Support and
Adherence
Through their ACE or PSM, patients can get help preparing for appointments, receive friendly reminders for monthly liver monitoring, and have their questions about AQVESME answered by text or phone.
Have your patients sign both the AQVESME REMS Patient Enrollment Form and the AQVESME Start Form while in the office, once counseled.
Your patients have a dedicated team at myAgios
Agios Clinical Educators (ACEs) and Patient Support Managers (PSMs) are here to help guide your patients living with thalassemia along every step of their journey.
What's an ACE?
An Agios Clinical Educator (ACE) is there from the start to offer personalized guidance to people living with thalassemia, with or without a prescription.
ACEs help your patients:

Learn about their disease, set goals for living with their condition, and discuss treatment and monthly liver monitoring requirements.

Get introduced to their PSM to learn more about health insurance coverage and access to AQVESME.

Build connections through webinars, community events, advocacy groups, and Facebook and Instagram pages.
What's a PSM?
A Patient Support Manager (PSM) guides patients in helping get their AQVESME prescription filled, qualify for financial assistance, get personalized support, and more.

PSMs help your patients:
- Connect to ACEs to learn about their condition and treatment considerations
- Understand financial assistance and eligibility criteria
- Get their AQVESME prescription filled
- Provide support through ongoing adherence

PSMs help you and your office:
- Ensure all forms are received and completed
- Navigate the patient's insurance coverage
- Address prior authorization and appeals requirements
- Coordinate all fulfillment and delivery with the exclusive REMS-certified Specialty Pharmacy
ACEs and PSMs do not provide medical advice. For medical advice or treatment-related questions, patients are instructed to talk to their healthcare team.
Is your patient not a native English speaker? No problem.
We have translation services available in the following languages: 简体中文 (Chinese), يبرع (Arabic), Tiếng Việt (Vietnamese), ودرا (Urdu), हिंदी (Hindi), and many more.
Start AQVESME in 3 simple steps
Once you and your patient have reviewed AQVESME clinical and REMS information and decided AQVESME is right for them, these are the steps to start treatment promptly:
Complete the AQVESME Start Form and your one-time HCP AQVESME REMS certification
After you and your patient sign and submit the AQVESME Start Form, the patient will be enrolled in myAgios® Patient Support Services. The myAgios team can assist you and your patient in determining insurance coverage and exploring financial assistance options.
Ensure your patient's pre-treatment liver test is completed
The pre-treatment liver test (blood test) must be completed within 4 weeks of the first prescription being filled. The myAgios team will work with you and your patient to determine the ideal timing to complete the required pre-treatment liver test based on the status of insurance determination.
Enroll your patient in AQVESME REMS
To enroll, you and your patient must complete and sign the AQVESME REMS Patient Enrollment Form, including documentation of the patient’s pre-treatment liver test and confirmation that the patient is appropriate for AQVESME.
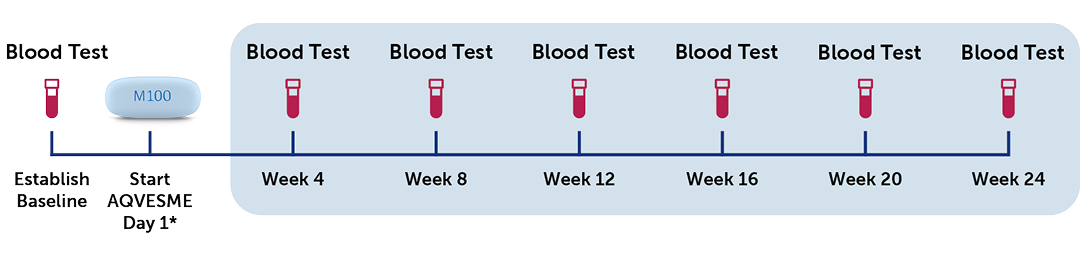
Patient monitoring requirements include a pre-treatment liver test before initiating therapy and monitoring liver function every 4 weeks during the first 24 weeks of treatment and as clinically indicated thereafter.
*AQVESME is one pill taken twice daily, supplied as a 28-day pack.
The myAgios team will help ensure AQVESME is delivered directly to your patient's home through an exclusive REMS-certified Specialty Pharmacy.
For any questions regarding REMS, visit www.AQVESMEREMS
.com or call
1-800-625-9951.
Financial support for your patients
myAgios can connect patients to financial assistance and coverage support programs to help with access to AQVESME.

Here are 3 ways our myAgios team helps your patients get, afford, and continue on AQVESME:

$0 responsibility for commercially insured patients, regardless of how much they make
This program lowers costs for eligible patients to $0 per prescription, if their prescription copay exceeds that amount. Support is available for those who qualify. Patients who have government insurance are not eligible for this program.
Please see full Terms and Conditions for more information.
We’re ready to help!
If you and your practice have any questions about patient eligibility, find your PSM for answers or call us at 1-877-77-AGIOS (1-877-772-4467).

Prescribe and connect your patients
The AQVESME Start Form is a single form that connects your patients to myAgios to get all the support they need.

Explore more
Find out more about efficacy, safety, and dosing for AQVESME.
We’re ready to help
If you and your practice have any questions about patient eligibility, find your PSM for answers.
Or call us at 1-877-77-AGIOS (1-877-772-4467).